Melamine combined with formaldehyde or urea produces a product called melamine resin. Melamine found in floor tiles, kitchenware and fire retardant fabrics is fire resistant and tolerant of high temperatures. Melamine resin, also known as thermoset plastic, begins as a white powder. Formica, made from melamine, provides a durable coating for shelving, countertops and furniture. Aside from its advantages, melamine has its share of disadvantages as well.
Food Contamination
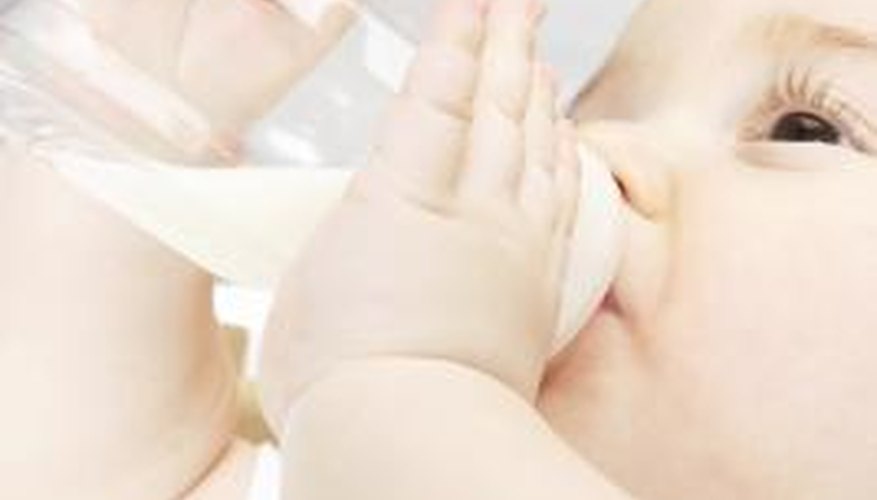
Baby formula laced with melamine caused alarm in the United States when over 49,000 children became ill in China. Imported baby formulas, powdered milk and other products imported from China contained high levels of melamine. Since melamine compounds mimic those of protein, various forms of the toxic substance added to foods raise the level of protein in the products and for some products, such as bags of rice, melamine pellets add weight to the product. The actions were an attempt by China's companies to reduce manufacturing and shipping costs of their products.
- Baby formula laced with melamine caused alarm in the United States when over 49,000 children became ill in China.
- Since melamine compounds mimic those of protein, various forms of the toxic substance added to foods raise the level of protein in the products and for some products, such as bags of rice, melamine pellets add weight to the product.
Plastic Containers
Many food containers and packaging products made from melamine leach the toxic substance into food. According to the International Herald Tribune, Government officials claim people can tolerate ingesting a small amount of the substance. The death of three children in China resulted in a set melamine limit in infant formula of 1 mg per kg of milk product. Although leaching from plastic containers is allowable, adding melamine to foods is now illegal and products from China are tested regularly.
- Many food containers and packaging products made from melamine leach the toxic substance into food.
- According to the International Herald Tribune, Government officials claim people can tolerate ingesting a small amount of the substance.
Melamine Paint
Melamine paint is durable, lasting 10 years or longer with proper care and maintenance. Although durable, it cuts easily with a sharp object and flakes when exposed to prolonged moisture. Do not use a brush to apply melamine paint; a foam roller provides a more evenly distributed coat of paint. Melamine paint requires one coat of primer and two coats of paint. The biggest disadvantage to this type of paint is its drying time; each coat takes at least a week to dry.
- Melamine paint is durable, lasting 10 years or longer with proper care and maintenance.
Melamine Foam
A block of melamine foam, called a magic eraser, erodes stains with its tiny solid fibres. Due to air pockets between fibres, melamine foam also makes a great soundproofing material by absorbing and blocking sound. Although great for cleaning, melamine foam sponges are more expensive than regular sponges and deteriorate quickly with use.