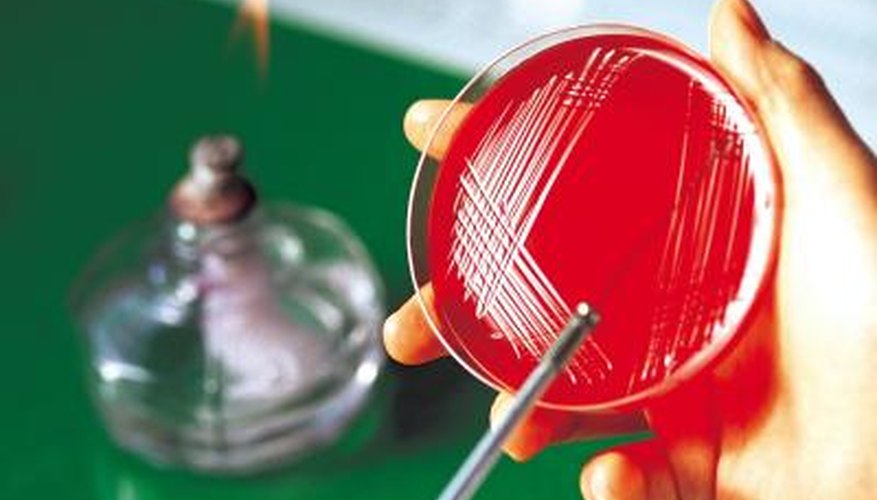
A growth medium, also known as a culture medium, is a liquid or gelatinous solid in, or on which microbiologists grow microorganisms. A major breakthrough in microorganism identification occurred when Robert Koch and his colleagues developed the techniques necessary to grow and isolate individual species of bacteria using solid media. Although the invention of solid media and its many advantages revolutionised the field of microbiology, it has some disadvantages.
Composition
When growing microorganisms, such as bacteria, one must provide them with environmental and nutritional conditions similar to those found in their natural habitat. A growth medium must supply all the nutritional requirements necessary for growth, including water, a source of carbon and energy, a nitrogen source, trace elements and growth factors. Common growth media ingredients include water, agar, meat extract, yeast extract, malt extract, peptone and casein hydrolosate.
Consistency
Growth media are classified in numerous ways, one of which is consistency. Liquid media, commonly referred to as broths, are available in bottles, flasks and test tubes. A solid medium is a liquid medium to which a solidifying agent, usually agar, has been added. Agar is composed of linked sugar molecules, called polysaccharides, and is obtained from some species of marine red algae. It melts at 100 degrees Celsius and solidifies at approximately 43 degrees Celsius. Solid media are used in the form of plates or slants. Plates are shallow, cylindrical containers with lids, called petri dishes, that contain solid media. Slants are made by pouring melted media into tubes and cooling them at an angle to create a long slanted growth surface. An agar concentration of 1.5 per cent is typical for plates and slants.
- Growth media are classified in numerous ways, one of which is consistency.
- Agar is composed of linked sugar molecules, called polysaccharides, and is obtained from some species of marine red algae.
Advantages
Solid media are useful when it is likely that a specimen contains a mixed bacterial population. When plates are properly inoculated, bacteria grow as distinct colony-forming units. This allows for presumptive identification based on a bacterial colony's appearance and permits preparation of pure subcultures. Early preliminary identification helps physicians make timely treatment selections. Pure subcultures make definitive bacterial identification and antimicrobial susceptibility testing possible. Bacterial contaminants are also readily apparent on solid media.
- Solid media are useful when it is likely that a specimen contains a mixed bacterial population.
- This allows for presumptive identification based on a bacterial colony's appearance and permits preparation of pure subcultures.
Disadvantages
Microorganisms have different oxygen requirements. Aerobic organisms require oxygen while anaerobic organisms survive only in its absence. Unlike liquid media, solid media does not support the simultaneous growth of aerobic and anaerobic bacteria. Consequently, duplicate plates must be inoculated, and incubated in both atmospheres. Solid media cannot accommodate large inocula volumes, typically those greater than 0.05ml. Because plate media are shallow and have large surface areas, they can dehydrate quickly and lose their ability to support growth within a few days if evaporation prevention measures are not taken.
- Microorganisms have different oxygen requirements.
- Because plate media are shallow and have large surface areas, they can dehydrate quickly and lose their ability to support growth within a few days if evaporation prevention measures are not taken.