Salbutamol is a widely used asthma medication sold under numerous trade names, including Ventolin and Proventil. It is a bronchodilator, meaning that it acts on specific cells in the airway to widen the air passages. Its mechanism of action, which is through interaction with a specific cellular receptor, the beta-2-adrenergic receptor (b-2AR), has been intensely studied. The binding of salbutamol to b-2AR "activates" the receptor, stimulating it to produce chemicals that eventually lead to relaxation of the airway muscles.
How It Works

Salbutamol is only one of many molecules that binds to and activates b-2AR. Epinephrine (adrenalin) is the natural binding partners for the receptor, and the neurotransmitter dopamine binds to it as well. Several other asthma medications are also b-2AR activators; the drugs known as beta blockers, including propranolol, bind to b-2AR but do not activate it. The varied effects of these molecules is due to differences in their functional groups and how they bind. B-2AR is a membrane protein that weaves its way from the internal to the external surface of the cell membrane seven times, forming seven very close "stitches" that form a ring. Salbutamol interacts with three of b-2AR's seven transmembrane domains (TMDs).
- Salbutamol is only one of many molecules that binds to and activates b-2AR.
- Several other asthma medications are also b-2AR activators; the drugs known as beta blockers, including propranolol, bind to b-2AR but do not activate it.
Overall Structure
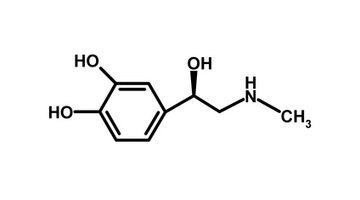
Salbutamol's molecular structure is similar to that of epinephrine (adrenalin), which was once also used as an asthma drug. Since adrenaline's structure binds with other receptors throughout the body, however, it caused unpleasant side effects.
Like adrenalin, the salbutamol structure is anchored at one end by a carbon ring with two pendant hydroxyl (OH) groups (called a catechol ring). Opposite of one hydroxyl group is a carbon chain with several more functional groups attached to it: another hydroxyl, an amine (NH), and a bulky hydrocarbon group called a "t-butyl" group. The t-butyl differentiates salbutamol from adrenalin and improves its specificity.
- Salbutamol's molecular structure is similar to that of epinephrine (adrenalin), which was once also used as an asthma drug.
- Like adrenalin, the salbutamol structure is anchored at one end by a carbon ring with two pendant hydroxyl (OH) groups (called a catechol ring).
Aromatic Ring

The carbon ring of salbutamol has alternating single and double bonds that give it a special electronic property known as "aromaticity." (Many, but not all, aromatic rings actually have characteristic odours, which is the origin of the name.) Aromatic rings are attracted to each other, and the ring of salbutamol is believed to fit into b-2AR near three other aromatic rings in TMD 5 and the extracellular region of the protein. This is a subtle difference from adrenalin, which binds to a phenylalanine amino acid in TMD 6.
Hydroxyls
The two hydroxyls directly connected with the aromatic ring fit into the b-2AR structure in TMD 5, attracted by three serine amino acids. Hydroxyls are not typically attracted by serines, which also contain hydroxyl groups, but are in this case due to the aromatic ring's special electronic properties. The hydroxyl on the tail nestles into TMD 6, attracted to a slightly positive charge on an asparagine amino acid located there.
Amine and t-Butyl Group

The amine group has a slight positive charge and is attracted to a negatively-charged aspartic acid amino acid in TMD 3. The bulky t-butyl group attached to the amine fits into the roomy pocket of this TMD and interacts with other non-polar amino acids inside. The b-2AR receptor can receive this extra bulk, whereas other receptors that adrenalin activates cannot. This is key to salbutamol's specificity and reduced side effects relative to adrenalin for asthma treatment.
- The amine group has a slight positive charge and is attracted to a negatively-charged aspartic acid amino acid in TMD 3.
- The bulky t-butyl group attached to the amine fits into the roomy pocket of this TMD and interacts with other non-polar amino acids inside.
