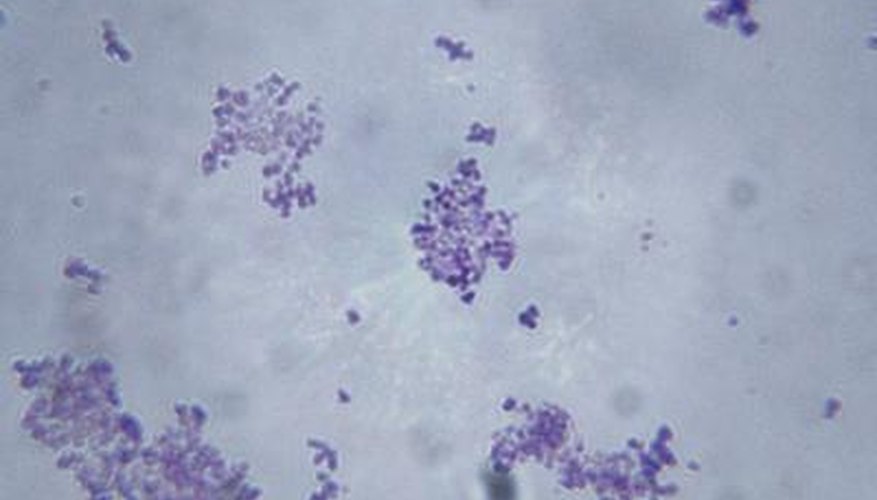
Microorganism identification plays a significant role in the treatment of disease. Microscopic examination of a stained smear is usually the first step in detecting and identifying disease-causing bacteria. Although the procedure does not make a definitive identification, it provides important clues regarding the organism's size, shape and structural composition. Stains are either direct or indirect. When a direct stain is used, it can be either simple or differential. Despite its limitations, direct staining has numerous advantages.
Smear Preparation
The first step in direct bacterial staining procedures is preparation of a smear. Cells from a culture or specimen are spread in a thin film over a glass microscope slide, dried and fixed using heat or a chemical fixative. Fixing the smear attaches the bacteria to the slide and prevents them from being washed away during the staining procedure. Smears should be thin enough to allow observation of individual cells and fixed sufficiently to allow repeated washings during the staining procedure.
- The first step in direct bacterial staining procedures is preparation of a smear.
- Fixing the smear attaches the bacteria to the slide and prevents them from being washed away during the staining procedure.
Stains
Because of their high water content, microbes are almost colourless. Staining enhances them and reveals their different characteristics. Stains are generally salts composed of charged particles, called ions. When mixed with water, a salt dissociates into positively charged cations and negatively charged anions. Stains are salts in which either the cations or anions are coloured. Depending on which type of ion carries the colour, common microbiological stains are either basic or acidic. Basic stains contain coloured cations and colourless anions. Examples of basic stains include methylene blue, basic fuchsin and crystal violet. Acidic stains have coloured anions and colourless cations. Examples include eosin, Congo red and nigrosin.
- Because of their high water content, microbes are almost colourless.
- Acidic stains have coloured anions and colourless cations.
Principle
Bacteria carry a slight net negative charge. Consequently, they attract and bind positively charged basic stains. Because the bacteria themselves retain the dye, the process is a direct staining method. A direct stain can be either simple or differential. Simple staining uses one dye to reveal the size, shape and arrangement of bacteria. A differential stain, such as the Gram stain, uses two different coloured basic dyes to distinguish types of bacteria in addition to their physical characteristics. Indirect, or negative staining techniques use acidic stains. The bacteria repel the dye and remain colourless against the stain-retaining background.
- Bacteria carry a slight net negative charge.
- A differential stain, such as the Gram stain, uses two different coloured basic dyes to distinguish types of bacteria in addition to their physical characteristics.
Advantages
Direct staining of bacterial cells is the most commonly used technique for visualising bacteria. It is inexpensive, simple and relatively fast. This technique provides health care professionals with same-day results, a potential life-saving advantage when diagnosing and treating some infections, such as bacterial meningitis. Direct stained smears are permanent preparations that can be re-examined at a later time, if necessary. Because direct staining kills the bacteria, your risk of infection from handling the smear is reduced.
- Direct staining of bacterial cells is the most commonly used technique for visualising bacteria.
- This technique provides health care professionals with same-day results, a potential life-saving advantage when diagnosing and treating some infections, such as bacterial meningitis.