Camphene, C10H16, is produced from alpha-pinene, and is a two-ringed monoterpene hydrocarbon. Terpenes are closely associated with conifers. In fact, the word "terpene" comes from "turpentine".
Camphor, C10H16O, a pleasant, medicinal-smelling terpenoid, once frequently found in the home, possesses antimicrobial and anesthetic properties. It is also used in cough-suppressants.
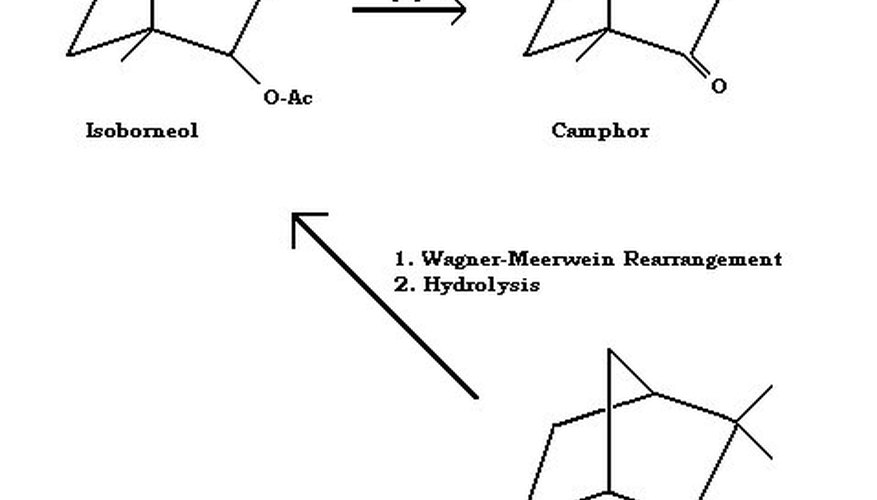
Camphor is made from camphene via intermolecular rearrangement, acetate capture, hydrolysis and then oxidation.
Reaction Sequence
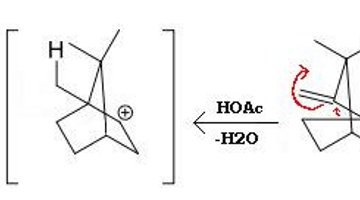
Camphene undergoes Wagner-Meerwein rearrangement, forming isoborneol by bond relocation. The diagram shows the three neutral species involved in the rearrangement.
Many reactions are reversible. By "capturing" one configuration, the reversibility is eliminated, and the desired product is produced in high yield. This is the purpose of the acetate in the synthesis of camphor--to drive the reaction in the desired direction--to the formation of isoborneol acetate. Sometimes this reaction is carried out in the presence of a catalyst, such as a tungsten-containing polyacid.
The isoborneol acetate may then be saponified to isoborneol by treatment with alcoholic potassium hydroxide. After cooling, the product is treated with water, causing the solid isoborneol, which is not water soluble, to precipitate. Collecting it by filtration and drying it, the isoborneol can then be oxidised using the mild oxidising agent, sodium hypochlorite bleach (in the presence of acetic acid). Alternatively, the oxidising mixture of chromic acid and sulphuric acid can be used, however, this generates troublesome chromium waste products.
- Camphene undergoes Wagner-Meerwein rearrangement, forming isoborneol by bond relocation.
- Collecting it by filtration and drying it, the isoborneol can then be oxidised using the mild oxidising agent, sodium hypochlorite bleach (in the presence of acetic acid).
Wagner-Meerwein Rearrangement and Carbocation Rearrangements
The carbocation (formerly called carbonium ion) was first observed in 1901. However, this rearrangement discovered by German chemist Hans Meerwein (1879-1965) was the first reaction proclaimed to have involved a carbocation intermediate.
Precautions
Some of the materials used in this synthetic procedure are potentially dangerous. Acetic acid, for instance, is capable of producing serious burns without inflicting much, if any, initial pain. In addition, the fumes are harmful to mucous membranes and the eyes. Alcoholic potassium hydroxide is strongly caustic. Even camphor and the other substances used as the major reactants are toxic and may cause problems if the proper safety equipment, including laboratory hoods, is not used.
- Some of the materials used in this synthetic procedure are potentially dangerous.
- Even camphor and the other substances used as the major reactants are toxic and may cause problems if the proper safety equipment, including laboratory hoods, is not used.