Titration is a process or method to determine the concentration of a substance in an unknown solution, in which a known reagent is added to an unknown solution to produce a known reaction such as a change in colour or electrical measurement. While it is more commonly associated with high school chemistry labs, medicine and the science industry, many common items use titration to test for the presence of various substances.
Blood Sugar Testing
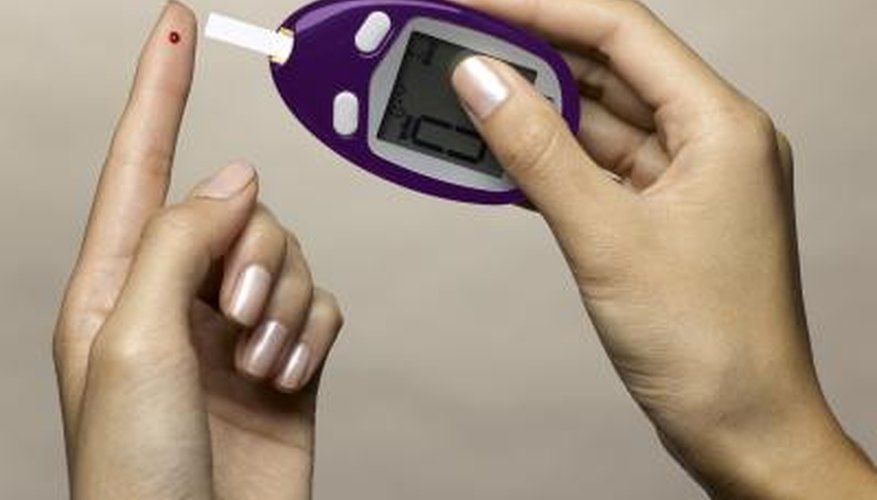
Diabetics can measure the amount of glucose in their blood by using a small portable machine called a blood glucose meter. To use the machine, a small sample of blood is applied to a test strip and mixed with reactants, then a small electrical current is applied to the sample. The current is affected by the concentration of the reactants and can then be used to measure the amount of glucose present in the blood.
Pregnancy Testing
Home pregnancy testing kits detect the presence of human chorionic gonadotrophin (hCG) in a subject's urine applied to a test strip or solution. The application of urine causes a colour change that will indicate either a positive or negative test result as early as two weeks after conception. A more comprehensive pregnancy test applies titration to a blood sample to measure the specific amount of hCG present in the blood. This test can also detect possible problems with the pregnancy and determine how long the subject has been pregnant and can only be performed by a health care provider.
- Home pregnancy testing kits detect the presence of human chorionic gonadotrophin (hCG) in a subject's urine applied to a test strip or solution.
- A more comprehensive pregnancy test applies titration to a blood sample to measure the specific amount of hCG present in the blood.
Aquarium Water Testing
The water in a home aquarium constitutes a small and delicately balanced environment for fish. If the properties of aquarium water change too drastically, disease and death of pets can result. In order to monitor changes in water conditions, home test kits are sold containing chemicals to test the pH level of water as well as the levels of ammonia, nitrites, nitrates and phosphates by placing a measured amount of the test chemical in a water sample and observing the extent, if any, of the resulting colour change.