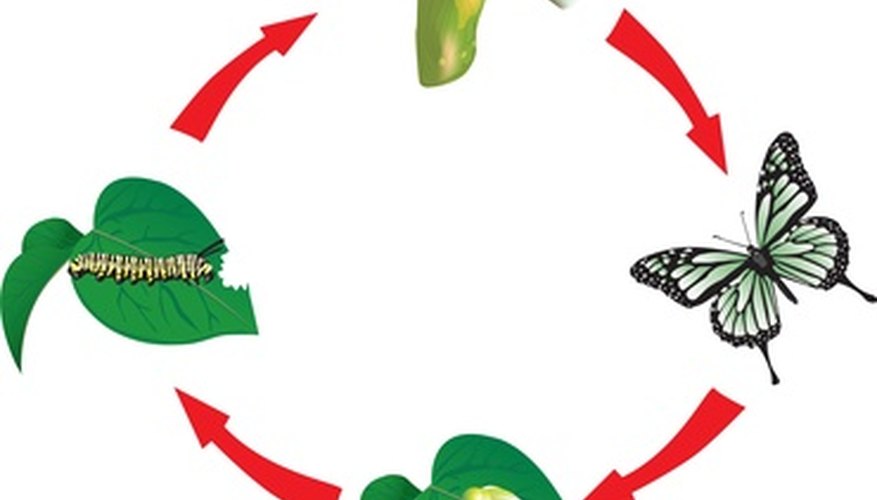
In order to teach children about the life cycles of living things, it is important to understand some of the misconceptions they start out with. They must understand that the requirements of a plant, for example, are similar but much different than the requirements of a butterfly. Exploring the facets of different life cycles gives children a firmer grasp of how living things must exist together for the benefit of all.
Breathing and Respiration

One of the most common misconceptions is the idea that all living things breathe in the same way. Respiration is a necessary function for all living, but it is accomplished in vastly different ways. Mammals have lungs and fish have gills, while trees perform respiratory functions through their leaves and many insects actually "breathe" through special openings in their thorax. Respiration is a common trait of all living things share, but it is a misconception that all life does it in the same manner.
- One of the most common misconceptions is the idea that all living things breathe in the same way.
- Respiration is a common trait of all living things share, but it is a misconception that all life does it in the same manner.
Animals and Nutrition

Many children mistakenly believe that all creatures eat the same foods or consume their foods in the same way. Learning about eating habits helps children understand the life cycle of organisms and how those life forms grow. Plants require nutrients in a chemical form, for instance, while more advanced life forms have stomachs that are able to convert raw ingredients into the necessary nutrients. Help them explore the relationship between very large creatures such as baleen whales and extremely small ones such as the krill they feed upon.
- Many children mistakenly believe that all creatures eat the same foods or consume their foods in the same way.
- Learning about eating habits helps children understand the life cycle of organisms and how those life forms grow.
Relationship of Plants and Animals

To children, the term "life cycle" is often interpreted to mean a direct chain of events. In truth, a more correct term wood be "food web" where many different life cycles interact. Insects and worms convert waste material into nutrients that can be used by plants that grow food that can be eaten by the people whose waste is later converted by insects. No form of life, no matter how small or large, exists without the support of many other types of life.
- To children, the term "life cycle" is often interpreted to mean a direct chain of events.
- No form of life, no matter how small or large, exists without the support of many other types of life.
Misconceptions About Reproduction

Most children understand that they came from their mother, and apply the mammal style of reproduction to other living things. Understanding that birds and snakes lay eggs, for example, helps children to learn about and recognise the different classes of animals such as birds and reptiles. Another example is that with very few exceptions, mammals do not lay eggs while very few reptiles are capable of giving live birth. Teach children how the way a species reproduces is directly tied to the environment that creature lives in.
- Most children understand that they came from their mother, and apply the mammal style of reproduction to other living things.
- Understanding that birds and snakes lay eggs, for example, helps children to learn about and recognise the different classes of animals such as birds and reptiles.